GROUPON GUIDE TO CHICAGO
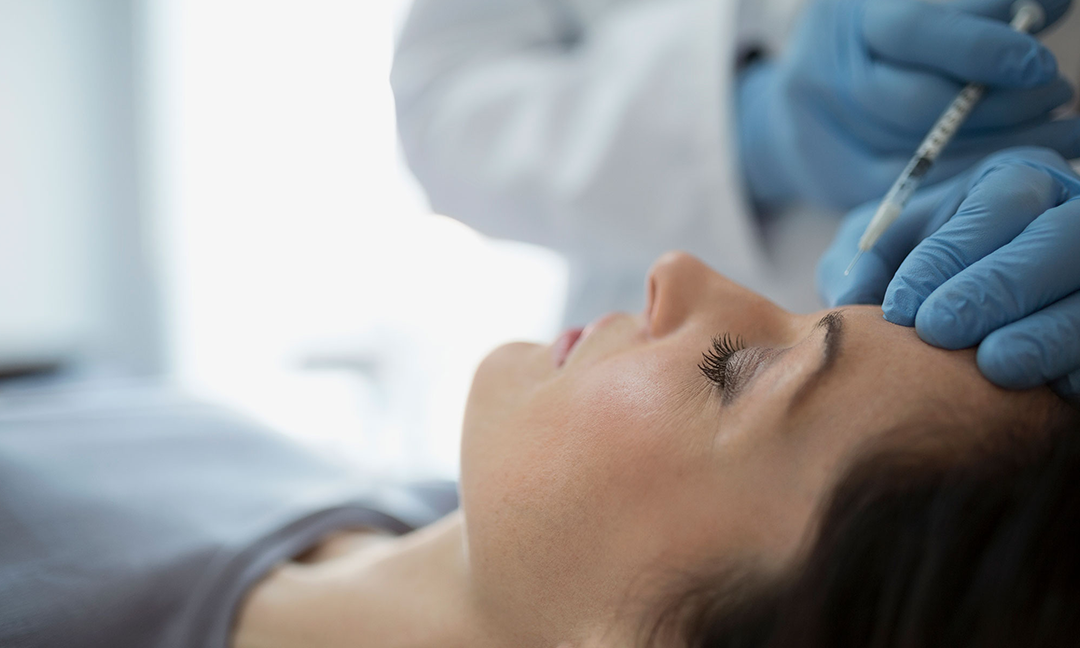
What Is Xeomin? A Guide to the Botox Alternative
BY: Colleen Loggins Loster |Nov 15, 2018
Deals on Xeomin

1.5 CC Radiesse & 20 Units Xeomin for a Youthful Look
1917 North Damen Avenue, Chicago • 3.7 mi
Sale Ends 5/12
$120.69 with_code SAVE
Ellehomme Wicker Park

Get Smoother Skin: 20 Units of Xeomin or 50 Units of Dysport Injection
3825 Highland Avenue, Downers Grove • 20.3 mi
Sale Ends 5/12
Metamorphosis Med Spa

Effortless Youth: 20 or 40 Units of Xeomin for Flawless Confidence
850 South Wabash Avenue, Chicago • 0.6 mi
Sale Ends 5/12
$181.44 with_code SAVE
LINZ Aesthetics Clinic

Achieve Wrinkle Reduction with 30 or 50 Units of Xeomin or Jeuveau
1136 South Delano Court, Chicago • 0.9 mi
43% discount_off
$190.40 with_code SAVE
Skinovatio Medical Spa South Loop

Revitalize Your Skin with 20 Units of Xeomin
875 North Dearborn Street, Chicago • 1.3 mi
Sale Ends 5/12
$120.69 with_code SAVE
Pure Med Spa

Experience Youthful Glow with 20 Units of Xeomin
233 East Erie Street, Chicago • 1.0 mi
36% discount_off
$133.12 with_code SAVE
Elle Homme Streeterville

Revitalize Your Look with 30 or 40 Units of Botox or Xeomin
935 Lakeview Parkway, Vernon Hills • 30.3 mi
Sale Ends 5/12
$328.54 with_code SAVE
Defined

Transform Your Skin with 20, or 40 Units of Xeomin
3135 West 111th Street, Chicago • 13.6 mi
Sale Ends 5/12
$121.50 with_code SAVE
Shaperz Inc

Rejuvenate with 20 Units of Xeomin Treatment
3409 North Kennicott Avenue, Arlington Heights • 26.1 mi
Sale Ends 5/12
$120.69 with_code SAVE
Artistic Touch Advanced Aesthetics

Smooth Wrinkles w/ 20 Units of Xeomin at Shape Med Spa by Dr. Katsaros
7863 Broadway, Merrillville • 31.6 mi
Sale Ends 5/12
$113.40 with_code SAVE
Dr. Katsaros, Shape Med Spa
Deals on Other Injections

One or Two Syringes of Restylane Fillers
875 North Dearborn Street, Chicago • 1.3 mi
Sale Ends 5/12
$387.99 with_code SAVE
Pure Medical Spa

Reduce the Appearance of Frown Lines w/ Up to 60 Units of Jeuveau
4256 North Ravenswood Avenue, Chicago • 6.1 mi
Sale Ends 5/12
$144.99 with_code SAVE
Ravenswood Health and Wellness Center

Boost Your Glow with Natural PRF Treatments
9933 Lawler Avenue, Skokie • 14.2 mi
29% discount_off
Northshore Health and Beauty

Up to 44% Off on at Dermaesthetics
9071 Foothills Boulevard, Roseville
44% discount_off
Dermaesthetics

Liquid BBL and Body Contouring Packages
520 South Michigan Avenue, Chicago • 0.3 mi
30% discount_off
$4,850 with_code SAVE
Beauty Magic lyadat sihr al jmaa

Premium Filler Enhancements
15000 South Cicero Avenue, Oak Forest • 18.9 mi
Sale Ends 5/12
$175.39 with_code SAVE
The Aesthetic Solution

1.5 CC Radiesse & 20 Units Xeomin for a Youthful Look
1917 North Damen Avenue, Chicago • 3.7 mi
Sale Ends 5/12
$120.69 with_code SAVE
Ellehomme Wicker Park

Up to 38% Off on Injection - Hyaluronic Acid at Midwest Surgical Arts
1250 North Mill Street Ste 103, Naperville • 28.0 mi
Sale Ends 5/12
$334.77 with_code SAVE
Midwest Surgical Arts

Achieve a Fresh Look with One Syringe of Revanesse Versa
875 North Dearborn Street, Chicago • 1.3 mi
Sale Ends 5/12
$347.49 with_code SAVE
Pure Medical Spa

Restylane Gel Sessions for Lips, Cheeks, and Contouring
520 South Michigan Avenue, Chicago • 0.3 mi
55% discount_off
$400 with_code SAVE
Beauty Magic lyadat sihr al jmaa

Get Smoother Skin: 20 Units of Xeomin or 50 Units of Dysport Injection
3825 Highland Avenue, Downers Grove • 20.3 mi
Sale Ends 5/12
Metamorphosis Med Spa

Effortless Youth: 20 or 40 Units of Xeomin for Flawless Confidence
850 South Wabash Avenue, Chicago • 0.6 mi
Sale Ends 5/12
$181.44 with_code SAVE
LINZ Aesthetics Clinic

Achieve Wrinkle Reduction with 30 or 50 Units of Xeomin or Jeuveau
1136 South Delano Court, Chicago • 0.9 mi
43% discount_off
$190.40 with_code SAVE
Skinovatio Medical Spa South Loop

Experience rejuvenation with one vial of Sculptra
875 North Dearborn Street, Chicago • 1.3 mi
Sale Ends 5/12
$579.10 with_code SAVE
Pure Med Spa